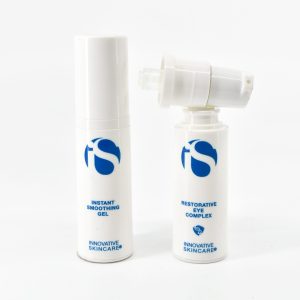
CR Nasal Spray Bottle
Child-Resistant | Medical-Grade | Metered Dosage
15ml, 30ml
SKU: 766HP-CRN-15
- Medical-grade HDPE construction — chemically resistant, FDA-compatible
- 16 CFR §1700.20 certified child-resistant closure
- 50–100mcl precision metered dose per actuation
- Available in 15ml (D: 28mm × H: 107mm) and 30ml (D: 34mm × H: 118mm)
- Recyclable HDPE — meets institutional sustainability requirements
- Custom color, labeling, and lot coding available
- Compliance documentation available on request
- Built for pharmaceutical manufacturers, medical cannabis programs, and clinical labs
- MOQ: 50,000 units
Description
Why Choose the CR Nasal Spray Bottle?
A medical-grade, child-resistant nasal spray format built for pharmaceutical companies, clinical labs, and established regulated brands that need precise metered dosing, certified CR compliance, and production-proven performance at scale.
Medical-grade HDPE construction
High-density polyethylene construction — chemically resistant, FDA-compatible, and built to the material standards expected by pharmaceutical and clinical production environments.
Certified child-resistant closure
16 CFR §1700.20 certified CR mechanism — meets federal child-resistant standards accepted by pharmaceutical regulators, cannabis medical programs, and regulated wellness distribution channels.
Precision metered dosing
50–100mcl metered dose per actuation — consistent, controlled delivery every spray. Critical for cannabinoid nasal formulas, pharmaceutical nasal applications, and any treatment where dosing accuracy directly impacts clinical outcomes.
Built for high-volume production
50,000 unit MOQ — engineered for established brands, pharmaceutical manufacturers, and contract fillers running large-scale production programs, not pilot batches.
Recyclable — eco-responsible
HDPE construction is fully recyclable — meeting sustainability standards increasingly required by institutional buyers, healthcare procurement teams, and regulated retail channels.
Customizable for your program
Custom color, labeling, and decoration available — so your nasal spray program can maintain brand and compliance presentation standards across every unit at scale.
Sourcing for a large-scale nasal program?
We'll confirm size, decoration specs, production lead time, and compliance documentation for your program.
MOQ: 50,000 units
CR Nasal Spray Bottle — Medical-Grade Metered Dosage Packaging
Production scale note
This product has a minimum order quantity of 50,000 units. It is designed for pharmaceutical manufacturers, contract packaging organizations (CPOs), licensed cannabis medical programs, clinical research facilities, and established regulated brands with high-volume production needs. If you are evaluating this product for a pilot or smaller initial run, contact our team to discuss options.
The CR Nasal Spray Bottle (SKU: 766HP-CRN-15) is a medical-grade, child-resistant nasal delivery format designed for pharmaceutical companies, clinical labs, licensed cannabis medical programs, and regulated wellness brands that require precision dosing, certified CR compliance, and production consistency across large-scale runs.
Constructed from high-density polyethylene (HDPE), the bottle delivers a metered dose of 50–100 microliters per actuation — ensuring consistent, controlled delivery on every spray. This level of dosing precision is critical for cannabinoid nasal formulas, pharmaceutical nasal applications, allergy and sinus treatments, saline and wellness solutions, and any clinical application where delivery consistency directly affects treatment outcomes or regulatory documentation.
The child-resistant closure is certified to 16 CFR §1700.20 standards, meeting federal CR requirements for pharmaceutical and regulated cannabis medical products. HDPE construction is chemically resistant, FDA-compatible, and fully recyclable — meeting the material and sustainability standards expected by institutional procurement teams and regulated distribution channels.
Available sizes
Standard clinical nasal format
Extended supply / higher volume programs
Who this product is built for
Pharmaceutical Manufacturers
OTC and Rx nasal spray programs requiring certified CR closures, metered dose consistency, and medical-grade material documentation.
Cannabis Medical Programs
Licensed medical cannabis operators producing nasal delivery formats for regulated medical dispensary and clinical distribution channels.
Clinical & Research Labs
Research facilities and contract fillers developing nasal delivery programs that require consistent metered dosing and CR-compliant packaging for study protocols.
Regulated Wellness Brands
Established wellness and nutraceutical brands running large-scale nasal spray programs — saline, allergy, sinus, and therapeutic nasal formats at institutional volumes.
Customization options
- Custom color on bottle body and cap — matched to brand or clinical program standards
- Label application — pressure-sensitive labels, shrink sleeves, and direct print options
- Custom actuator color and nozzle tip options available for program differentiation
- Lot number, batch coding, and regulatory panel layouts supported
- Pre-production sampling and fill testing available before full production commitment
- Compliance documentation available on request for regulatory submissions
Common questions
Frequently asked questions
Is this nasal spray bottle truly child-resistant?
Yes. The closure is certified to 16 CFR §1700.20 child-resistant standards — the federal standard accepted by pharmaceutical regulators and cannabis medical program oversight bodies. Always verify state and program-specific CR requirements with your regulatory affairs team before production.
What is the metered dose output per actuation?
The metered pump delivers 50–100 microliters (mcl) per actuation — a consistent, controlled dose on every spray. Exact output can be confirmed during pre-production sampling and fill testing for your specific formula and viscosity.
What formulas are compatible with this bottle?
The HDPE construction is compatible with aqueous nasal solutions, saline formulas, cannabinoid nasal suspensions, pharmaceutical nasal sprays, allergy and sinus treatments, and other water-based topical nasal delivery formulas. Compatibility testing with specific formulas is recommended prior to full production — our team can support this during the pre-production phase.
Why is the MOQ 50,000 units?
This product is manufactured to medical-grade production standards with tooling, testing, and quality control processes that are cost-justified at 50,000 units and above. It is designed for established pharmaceutical, clinical, and regulated cannabis medical programs with validated production volumes. If you are earlier in your program development, contact our team to discuss your timeline and requirements.
Is compliance documentation available?
Yes. Compliance documentation — including CR certification, material specifications, and testing data — is available on request to support regulatory submissions, product dossiers, and institutional procurement requirements. Contact our team to discuss your specific documentation needs.
Can I get samples or conduct fill testing before committing to full production?
Yes. We support pre-production sampling and fill compatibility testing before full production commitment. This is strongly recommended for pharmaceutical and clinical programs to confirm dosing consistency, formula compatibility, and decoration specifications before ordering at volume.
The packaging infrastructure medical and pharmaceutical programs demand
Medical nasal spray packaging operates in a higher-stakes environment than consumer topicals. Every aspect of this bottle — material, closure, dosing mechanism, and documentation — is engineered to meet the expectations of pharmaceutical procurement, regulatory review, and institutional quality assurance.
16 CFR §1700.20 CR certified
Federal child-resistant standard accepted by pharmaceutical regulators and medical cannabis program oversight bodies across major U.S. markets.
Medical-grade HDPE material
Chemically resistant, FDA-compatible HDPE construction — the material standard expected by pharmaceutical manufacturers and clinical production facilities.
Precision metered dosing
50–100mcl per actuation — consistent controlled delivery for clinical applications where dosing accuracy affects treatment outcomes and regulatory documentation.
Documentation support
CR certification, material specs, and testing data available on request — supporting regulatory submissions, product dossiers, and institutional procurement processes.
Specifications
| SKU | 766HP-CRN-15 |
| Available sizes | 15ml · 30ml |
| Dimensions — 15ml | D: 28mm × H: 107mm |
| Dimensions — 30ml | D: 34mm × H: 118mm |
| Material | HDPE (medical-grade, FDA-compatible) |
| Metered dose | 50–100mcl per actuation |
| CR certification | 16 CFR §1700.20 child-resistant |
| Recyclability | Recyclable HDPE |
| MOQ | 50,000 units |
| Use case | Pharmaceutical nasal sprays, medical cannabis nasal delivery, clinical research, saline and allergy treatments, regulated wellness nasal programs |
| Documentation | CR certification, material specs, and testing data available on request |
Related packaging
Building a full CR topical or medical packaging program? Here are the most commonly paired formats.