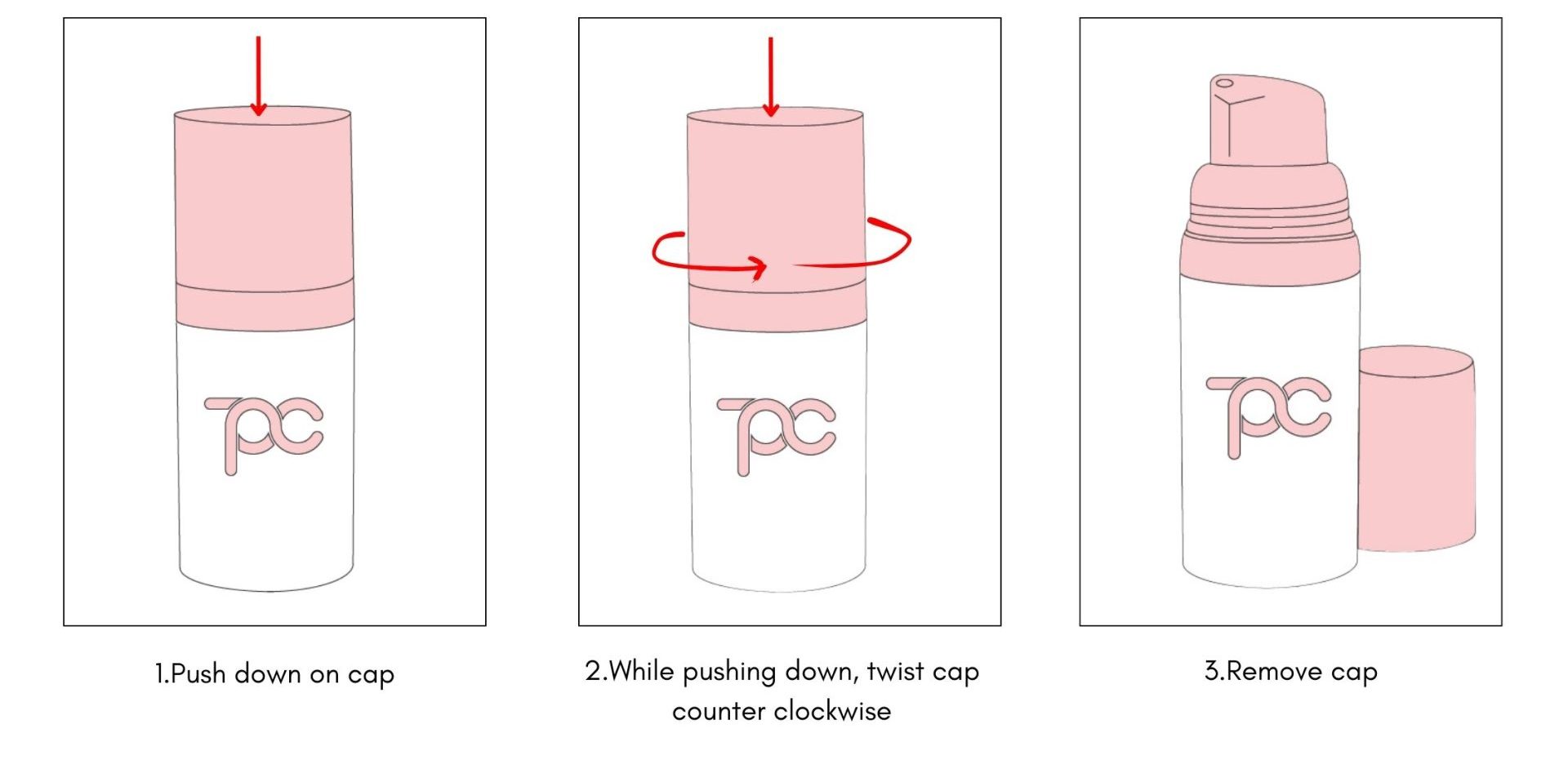
Child-Resistant Mono-Material Airless Bottle
Recyclable | Airtight | Child-Resistant
15ml, 30ml, 50ml
SKU: 144AB-GR249
Minimum Order Quantity: 10,000
- 100% recyclable mono-material
- 16 CFR §1700.20 Child-Resistant Certified closure
- Airtight airless system to protect active formulations
- 0.21ml precision pump for controlled dosing
- Reduces oxidation and contamination risk
- Custom decoration and scalable production options
Description
Airtight, Child-Resistant Airless Bottle — Now in a Mono-Material Format
A production-ready airless bottle built for regulated topical programs that need an airtight seal, a child-resistant certified closure system, and a simplified material architecture designed to support recyclability goals.
Why teams choose this format
Designed for predictable production, clean dispensing, and a premium retail presence — with a material approach that supports EU EPR/PPWR direction.
- Cleaner dispensing: sealed airless system helps reduce contamination risk.
- More consistent performance: airless dosing supports repeatable user experience.
- Simplified recyclability story: mono-material architecture supports sorting + recovery.
- Brand-ready: decoration options to maintain premium shelf appeal.
Quick specs buyers look for
Elevate your topicals with our CR Mono-Material Airless Bottle
Built for compliance-driven topical programs that want product protection, safer dispensing, and a recyclability-forward material approach.
| Item Number | 144AB-GR249 |
| Name | CR Mono Material Airless Bottle |
| Minimum Quantity | 10,000 |
| Material | Polypropylene / PCR / PP |
| Capacity | 15ml, 30ml, 50ml |
| Dimension | |
| Color & Finish | In-Mold Color, UV Printing, & Silk Screen Printing |
Common Questions
Is this airless bottle airtight?
What makes it “mono-material” and why does it matter?
Can we customize decoration and finishes?
What’s the minimum order quantity and typical lead time?
Built for Regulated, Compliance-Driven Topical Programs
Packaging decisions in regulated categories can’t be “close enough.” This format is designed to support safer dispensing, predictable performance, and cleaner handoffs from sampling into full-scale production.